This document outlines the importance of standardized documentation in clinical research and focuses on the crucial role of the Case Report Form Template Clinical Trials. Accurate and consistent data collection is paramount to the integrity of any clinical trial, and a well-designed case report form (CRF) is the cornerstone of this process. These forms are used to systematically record all relevant information about a patient’s participation in a clinical trial, from demographics and medical history to treatment details and adverse events. Choosing or creating the right Case Report Form Template Clinical Trials can significantly impact data quality, reduce errors, and ensure regulatory compliance.
Effective clinical trials rely heavily on meticulous data collection. The Case Report Form Template Clinical Trials serves as the primary tool for gathering this data, ensuring uniformity across all study participants. Without a standardized format, inconsistencies can arise, making it difficult to analyze results and draw valid conclusions. This article will delve into the essential components of a robust CRF, the benefits of using templates, considerations for design and implementation, available options, and best practices for ensuring data integrity.
A Case Report Form (CRF) is a standardized form used to collect data from patients participating in a clinical trial. It acts as a structured questionnaire or electronic form that captures all relevant information about a patient’s condition, treatment, and outcomes. Think of it as the primary source document for all data related to an individual participant. CRFs are indispensable for:

The core function of a CRF is to ensure consistency in data collection. By using a pre-defined format, it minimizes variations in how data is recorded, leading to more reliable and comparable results. This standardization is crucial for statistical analysis and drawing meaningful conclusions from the trial.
Well-designed CRFs incorporate validation checks to reduce errors and omissions. These checks can range from simple range limitations to complex logic tests that ensure data is accurate and complete.
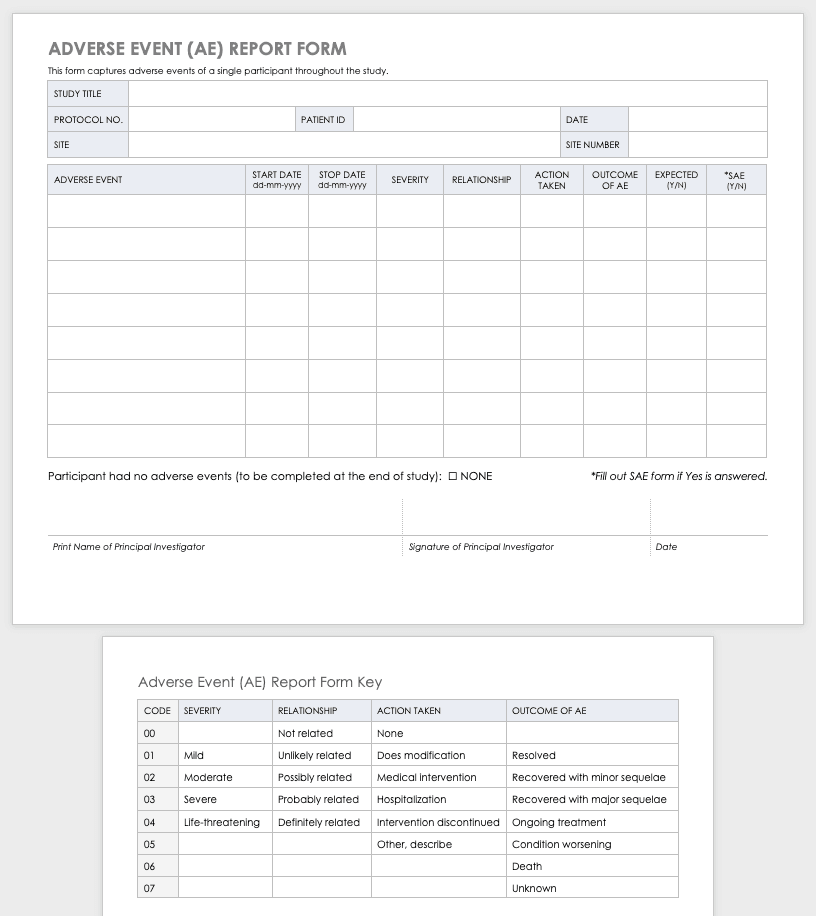
Regulatory bodies such as the FDA and EMA require robust data collection practices. CRFs play a vital role in demonstrating compliance with these regulations by providing a clear audit trail of data collection. The CRF itself becomes a critical component of the clinical trial’s documentation.

Accurate and timely data collection is essential for monitoring patient safety and identifying potential adverse events. CRFs facilitate the recording of adverse events and other safety-related information, enabling prompt intervention and appropriate management.

Developing a CRF from scratch can be time-consuming and complex. Fortunately, many resources offer pre-designed Case Report Form Template Clinical Trials that can significantly streamline the process. Utilizing a template offers several advantages:
Templates provide a ready-made framework, reducing the time and effort required to design a CRF. This is especially beneficial for researchers who are new to CRF development.
Well-designed templates often incorporate best practices and validation checks, minimizing the risk of errors and omissions. Many templates are developed by experienced CRF designers and incorporate lessons learned from previous trials.
By promoting standardization and incorporating validation checks, templates contribute to higher data quality, leading to more reliable results.
Using a template can be more cost-effective than hiring a CRF design specialist, particularly for smaller studies with limited budgets.
Reputable template providers ensure that their templates comply with relevant regulatory requirements, reducing the risk of non-compliance.
A comprehensive Case Report Form Template Clinical Trials typically includes several essential components. These components are designed to capture all relevant information about a patient’s participation in the trial.
This section captures basic information about the patient, such as age, gender, race, and contact details. This information is essential for identifying and tracking participants and for stratifying data analysis.
A detailed medical history section is crucial for understanding the patient’s pre-existing conditions and potential risk factors. This includes information on past illnesses, surgeries, medications, and allergies.
This section contains data related to the specific clinical trial, such as the assigned treatment, dosage, and duration of treatment.
A comprehensive AE section is essential for monitoring patient safety. This section captures all adverse events experienced by the patient, including their severity, duration, and relationship to the study treatment.
Regular measurement of vital signs such as blood pressure, heart rate, and temperature is a standard component of clinical trials. This section allows for the systematic recording of these measurements.
Laboratory tests are often performed to monitor the patient’s response to treatment and to assess potential side effects. This section allows for the recording of laboratory results and their interpretation.
This section captures data related to the efficacy of the study treatment. This may include measurements of disease severity, symptom scores, and other relevant outcome measures.
Traditionally, CRFs were paper-based documents. However, the rise of electronic data capture (EDC) systems has led to a significant shift towards electronic CRFs.
Paper-based CRFs are simple to implement and require minimal training. However, they are prone to errors, difficult to analyze, and time-consuming to manage. Data entry is also more labor-intensive.
EDC systems allow for the electronic collection and management of data. Electronic CRFs offer several advantages over paper-based CRFs, including:
* Improved data accuracy
* Reduced data entry errors
* Faster data analysis
* Enhanced data security
* Real-time data monitoring
* Automated validation checks
Popular EDC systems include Medidata Rave, Oracle Clinical, and Veeva Vault. The choice between paper and electronic CRFs depends on the complexity of the study, the budget, and the resources available. Many modern Case Report Form Template Clinical Trials are designed for use within EDC systems.
Designing an effective CRF requires careful planning and consideration of various factors. Here are some best practices for designing CRFs that are easy to use, minimize errors, and facilitate data analysis:
Use clear, concise language and avoid jargon. The CRF should be easy to understand for all users, regardless of their level of experience.
Organize the CRF in a logical sequence, grouping related questions together. This will make it easier for users to navigate the form and avoid missing information.
Incorporate validation checks to minimize errors and ensure data integrity. This may include range limitations, data type restrictions, and logic tests.
Design the CRF with the user in mind. Use appropriate formatting, such as spacing and shading, to improve readability.
Before implementing the CRF, pilot test it with a small group of users to identify any potential problems. This will help to ensure that the CRF is easy to use and that it captures all the necessary information.
Numerous resources offer pre-designed Case Report Form Template Clinical Trials. Here are a few options:
Many CROs offer CRF templates as part of their services. These templates are often customized to meet the specific needs of the study.
Specialized CRF development companies offer a wide range of pre-designed templates. These templates are typically designed for specific therapeutic areas or study types.
Some pharmaceutical and biotechnology companies make CRF templates available to the public. These templates may be useful for smaller studies or for researchers who are new to CRF development.
Several open-source resources offer free CRF templates. While these may require more customization, they can be a cost-effective option.
The Case Report Form Template Clinical Trials is a fundamental element in ensuring the quality, integrity, and regulatory compliance of clinical research. By understanding the benefits of using templates, the key components of a CRF, and best practices for design and implementation, researchers can streamline data collection, reduce errors, and improve the overall success of their clinical trials. The shift towards electronic CRFs has brought significant improvements in data management and analysis. Choosing the right template, whether paper-based or electronic, is crucial for achieving reliable and meaningful results. Investing in a well-designed and implemented CRF is an investment in the validity and credibility of clinical trial data.
In conclusion, effective data collection is a cornerstone of successful clinical trials, and the Case Report Form Template Clinical Trials plays an indispensable role in achieving this. Standardization, data integrity, and regulatory compliance are all enhanced through the use of well-designed and thoughtfully implemented CRFs. Whether utilizing a pre-designed template or creating a custom form, careful consideration of patient demographics, medical history, adverse events, and efficacy assessments is paramount. The move toward electronic data capture further strengthens the efficiency and reliability of CRF processes. By adhering to best practices and leveraging available resources, researchers can ensure that clinical trial data is accurate, complete, and readily analyzable, ultimately contributing to advancements in medical knowledge and patient care.